The Electron Microscopy Research Group (EMRG) generate ultrastructural and immunocytochemical data from surgical, biopsy tissue and cell culture samples at a resolution typically of 0.5 nm. Electron Microscopy is particularly valuable for visualising pathological changes and structures at the sub-cellular or organelle level and is traditionally used for the delineation of disease pathogenesis. Cryogenic sample fixation has enabled studies of single proteins and viral substructures at near-atomic resolution in structural biology.
Electron Microscopy platforms also may be configured for (i) 3D and wide area 2D studies (ii) elemental analysis using technologies such as Energy Dispersive Spectroscopy (EDS), (iii) spatial biomarker quantification and (iv) correlative light and electron microscopy (CLEM). The EMRG is a longstanding collaboration between the Ingham Institute, NSW Health Pathology, Western Sydney University and the Mark Wainwright Analytical Centre.
Our goals
The goal of the Electron Microscopy Research Group (EMRG) is to develop new approaches to the visualisation of cells and subcelluar fine structure for pathology diagnostics and cell biology research. These new approaches can embrace novel fixation and sample preparation protocols such as freeze substitution, nanoparticle immunocytochemical markers for correlation studies, computer-based quantitative image analysis, 3D structural rendering and new electron column designs such as for low kV STEM desktop systems. Much of this work is done at the Ingham Institute and includes:
- The development of array tomography for 3D ultrastructural imaging of adenocarcinoma of the prostate. Regions of interest are segmented either manually or using machine learning in order to understand cell-cell and cell-stroma interactions. 3D molecular tomography requiring more elaborate specimen preparation and specialised EM platforms such as available at the Mark Wainwright Analytical Centre are envisaged.
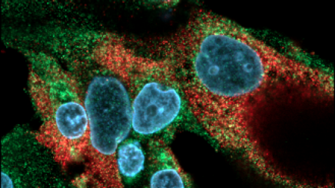
- The use of nanoparticle probes for immunocytochemical studies of cancer biomarkers and patient-derived circulating tumour cells (CTCs). Quantum dot nanoparticles have been extensively characterised and are used for multiplex immunostaining at a resolution approaching 15 nm.
Research strengths
- Strong clinical focus. The Ingham Institute is based in the Liverpool Hospital campus adjacent the NSW Health Pathology Centre/Liverpool Hospital and is therefore well positioned to be involved in translational clinical research.
- EM approaches are developed and optimised for human pathology tissue and cell culture samples.
- Strong historical linkage to the Electron Microscope Unit, Mark Wainwright Analytical Centre. This collaboration is fundamental for our ongoing work in specialised sample preparation, accessing state-of-the-art Electron Microscopy platforms and developing new nanoparticle probes.
- Our group knowledge of human disease pathogenesis combined with the underlying principles of cell biology, material science and nanotechnology allow strong interpretive skills in ultrastructure.
Our results
- The EMRG has pioneered the use of nanoparticle-based immunocytochemistry for multiplex immunostaining of fresh and archival human pathology samples.
- Our nanoparticle-based immunocytochemical technology has recently been expanded for use in correlative light and electron microscopy (CLEM).
- 3D array tomography feasibility studies have demonstrated the utility of this approach not only for understanding the tumour cell-matrix interface but also for conducting correlative studies with clinical imaging modalities such as MRI.
Our experts
Our affiliated partners
External links and affiliations:
- Ingham Institute for Applied Medical Research, Liverpool NSW
- SPHERE Cancer CAG
- Electron Microscope Unit, Mark Wainwright Analytical Centre
References
- Cohen-Hyams T, Mam K, Killingsworth MC. Scanning electron microscopy as a new tool for diagnostic pathology and cell biology. Micron 2020;130:102797
- Bhaskar S, Saab J, Cappelen-Smith C, Killingsworth M, Wu XJ, Cheung A, Manning N, Aouad P, McDougall A, Hodgkinson S, Cordato D. Clot histopathology in ischemic stroke with infective endocarditis. Canadian Journal of Neurological Sciences. 2019 May;46(3):331-336.
- Balaratnasingham C, Cherepanoff S, Dolz-Marco R, Killingsworth MC, Chen FK, Mendis R, Mrejen S, Curcio CA, Too LK, Gal-Or O, Freund KB, Yanuzzi LA. Cuticular drusen: Clinical phenotypes and natural history defined using multimodal imaging. Ophthalmology 2018 Jan;125(1):100-118
- Killingsworth, M.C., Bobryshev, Y.V. Correlative light- and electron microscopy using quantum dot nanoparticles. J. Vis. Exp. 2016 (114), e54307, doi:10.3791/54307.
- Killingsworth MC, Lai K, Wu X, Yong JLC, Lee CS. Quantum dot immunocytochemical localization of somatostatin in somatostatinoma by widefield epifluorescence, super-resolution light and immunoelectron microscopy. Journal of Histochemistry and Cytochemistry 2012;60(11):832 – 843



