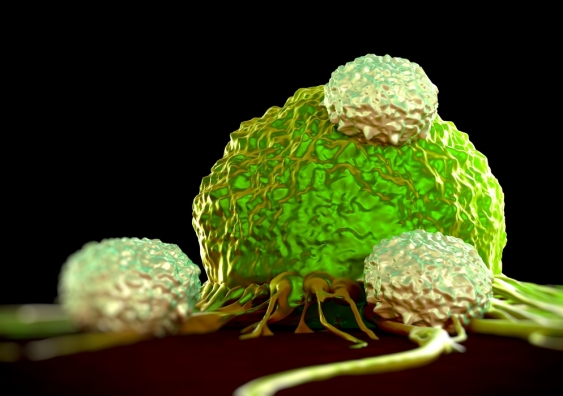
Your body’s cells use and resist force, and they move. It’s mechanobiology
New technology is revealing the mechanical aspects of cells and mechanobiology is enabling the development of new drugs and treatments, write Joshua Chou, Kate Poole and Maté Biro.